 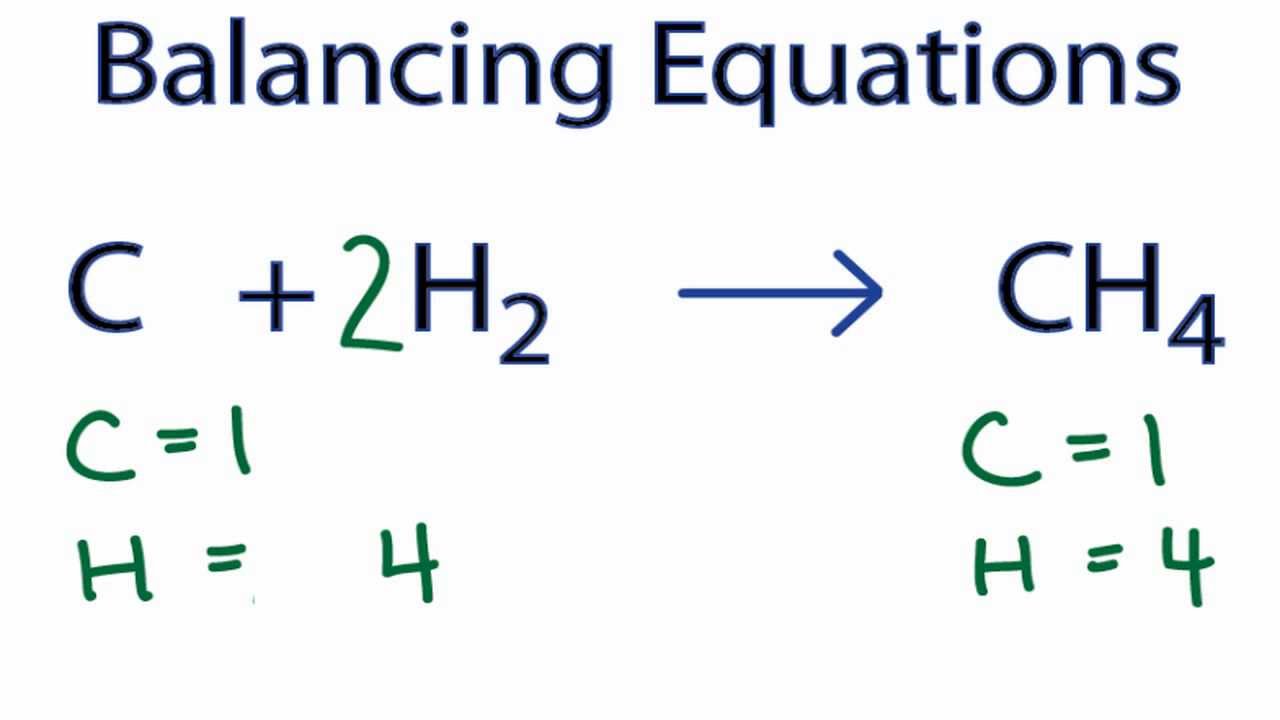
Let us try the same with another example.Ĭonsider the equation, KMnO 4 + HCl = KCl + MnCl 2 + H 2O + Cl 2įirst table the number of elements on each side to know the imbalance. Now, the equation is balanced with 4 Hydrogen's (H) and 4 Oxygen's on both sides. In order to balance the same, we add an another 2 in front of Hydrogen on the L.H.S (2) H 2 + O2 -> (2) H 2O 
Now, we have 2 Oxygen's but 4 Hydrogen's on the R.H.S. To balance this the unbalanced oxygen molecules, we add 2 in front of the product on R.H.S. Let us learn here how to balance the above unbalanced equation with step by step procedure. How to balance an unbalanced chemical equation? chemical equation by inspection methodIn this video, Ill cover step by step on how to balance chem. Hence with the unequal number of oxygen molecules the chemical equation is said to be unbalanced. Chapter 1: Matter1.3 Stoichiometrya) Write and balance i. Example: H 2 + O 2 -> H 2OĬonsidering the equation above, we have 2 hydrogen (H) and 2 oxygen (O) on the L.H.S and 2 hydrogen (H) and only 1 oxygen (O) on the R.H.S. Llly, an equation is said to be unbalanced when the number of molecules for each elements are not equal on both sides. In the above equation we have, 2 Sodium (Na) and 2 Chlorine (Cl) on both L.H.S and R.H.S, Hence the equation is said to be balanced. It is defined as the representation of reactions taking place between chemicals, with the reactants on the left hand side (L.H.S) and the product obtained from the reaction on the right hand side (R.H.S) When is an equation called balanced / Unbalanced?Īn equation is said to be balanced when there are equal number of molecules for each elements on both sides of the equations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
